AI-Powered APQR Software
Annual Product Quality Reviews, Reimagined with AI
Annual Product Quality Review is a critical regulatory requirement for pharmaceutical and life sciences organizations. AmpleLogic’s APQR solution enables structured, compliant, and inspection-ready reviews by consolidating quality data, automating trend analysis, and generating audit-ready reports.
Trusted by 100+ Pharma Companies Globally








Built to Support Modern Regulatory Expectations
Centralized APQR Data Management
Automatically consolidate deviations, CAPA, complaints, OOS, OOT, stability, batch, and manufacturing data into a centralized APQR platform for complete visibility, traceability, compliance oversight, and streamlined PQR management.
Accurate, Inspection-Ready Documentation
Ensure standardized templates, structured data capture, automated workflows, and complete audit trails to maintain data integrity, support regulatory compliance, and simplify global health authority inspections.
Seamless Cross-Functional Collaboration
Empower quality, manufacturing, quality control, and regulatory teams to securely collaborate in real time through a unified digital workspace, improving review efficiency and decision-making.
Secure, Controlled Audit Access
Empower quality, manufacturing, quality control, and regulatory teams to securely collaborate in real time through a unified digital workspace, improving review efficiency and decision-making.
Always Inspection-Ready with AmpleLogic APQR
Annual Product Quality Review (APQR) is a mandatory regulatory requirement in the pharmaceutical industry that ensures consistent product quality, robust process control, and continuous compliance with global regulatory standards such as FDA, EMA, and ICH guidelines. However, conventional APQR processes are often fragmented, time-consuming, and highly dependent on manual data collection, static statistical tools, and disconnected systems. This leads to extended review timelines, increased rework, limited data-driven insights, and elevated compliance and audit risks.
AmpleLogic’s Electronic APQR & CPV Solution addresses these challenges by converting traditional APQR into a centralized, automated, and inspection-ready digital quality management workflow. The platform seamlessly integrates data from LIMS, ERP, QMS, and other enterprise systems, creating a single, reliable source of truth for product quality reviews. Automated statistical analysis, dynamic control charts, and end-to-end traceability eliminate manual charting, reduce errors, and ensure data integrity and reproducibility.
Designed with regulatory compliance at its foundation, AmpleLogic APQR enables comprehensive APQR lifecycle management, covering data ingestion, trend analysis, deviation review, CAPA tracking, and electronic approvals. Built-in CPV monitoring, AI-driven summaries, and configurable dashboards allow quality teams to shift focus from manual report preparation to proactive risk management, continuous process improvement, and faster, evidence-based decision-making.
Supporting multi-site pharmaceutical operations with harmonized global templates and audit-ready documentation, AmpleLogic APQR helps organizations reduce APQR review timelines by up to 85%, improve inspection readiness, strengthen compliance posture, and gain real-time visibility into product and process performance across the enterprise.
Enhancing APQR with Intelligent Automation
Annual Product Quality Reviews involve large volumes of structured and unstructured data from multiple systems and departments.
Generate Reports Anytime
Generate APQR and PQR reports on demand using predefined, configurable templates with automated data population and analytics.

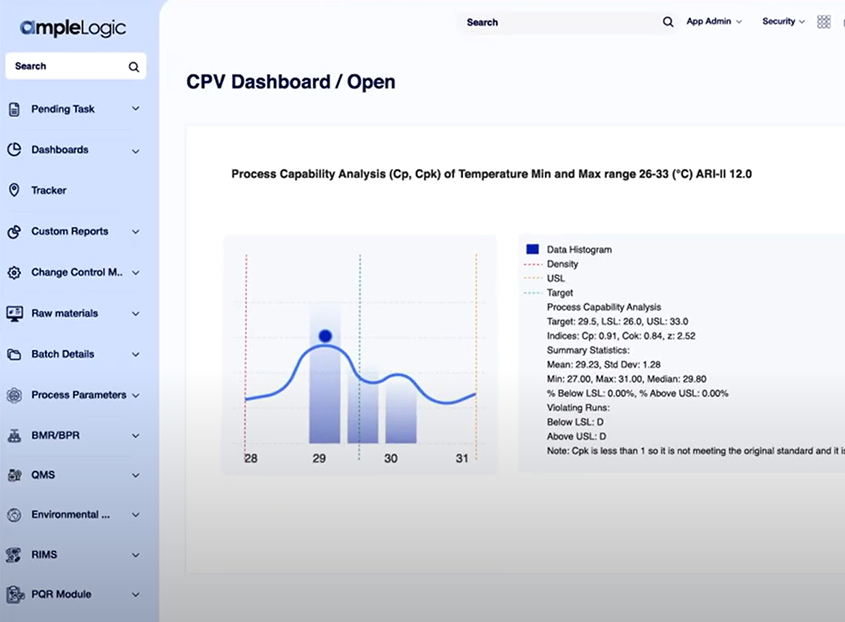
Continued Process Verification (CPV)
Enable continuous, automated monitoring of critical process and quality parameters across the manufacturing lifecycle with built-in statistical analysis and alerts.

OCR Data Extraction & AI Chatbot
Automatically extract quality data from physical documents, scanned records, and PDFs using OCR, and instantly access APQR and CPV insights through an AI-powered chatbot.

Key Functional Capabilities

Automated APQR / PQR Report Generation
Generate APQR, PQR, and Six-Pack reports on demand using standardized, configurable templates aligned with site-specific SOPs.

End-to-End Product Quality Review Automation
Streamline the entire APQR lifecycle from data collection to final approval, reducing manual effort and review timelines.

Audit Trails & Version Control
Maintain automatic version history, electronic records, and complete audit trails to ensure full traceability during inspections.

Global Regulatory Compliance
Ensure compliance with 21 CFR Part 11, EU Annex 11, MHRA, CDSCO, TGA, Health Canada, ANVISA, SFDA, and other global authorities.

Advanced Statistical Calculations
Automatically calculate Cp, Cpk, CPU, CPL, UCL, LCL, standard deviation, mean, and median for robust process evaluation.

Built-In Control Charts & Trend Analysis
Generate I Charts, Moving Range Charts, Normal Probability Plots, and Process Capability reports with automated statistical trending.

Nelson Rule & OOT/OOS Detection
Apply Nelson's Rules to detect non-random patterns and identify Out-of-Trend (OOT) and Out-of-Specification (OOS) conditions early.

Continued Process Verification (CPV)
Continuously monitor Critical Process Parameters (CPPs) and Critical Quality Attributes (CQAs) to maintain a sustained state of control.

Golden Batch & Process Capability Insights
Identify Golden Batch conditions through historical analysis and provide color-coded Cpk indicators for quick capability assessment.

Seamless System Integration
Integrate with LMS, QMS, DMS, MES, eBMR, and other enterprise systems for complete, end-to-end quality data capture.

OCR-Based Data Extraction
Digitize manual BMRs, scanned records, and PDFs using OCR to accelerate APQR generation and improve data accuracy.

Centralized Monitoring, Alerts & AI Assistance
Maintain centralized master data with defined limits, trigger automated alerts for deviations and review delays, and use an AI chatbot for instant APQR data retrieval and faster audit interactions.
Built-In Insights Without External Tools
AmpleLogic APQR includes integrated analytics and dashboards to visualize quality trends, risks, and performance indicators — eliminating dependency on external reporting tools.
Modules
Navigating Excellence Through Integrated Modules
Continued Process Verification
Pharmaceutical manufacturing in the modern pharmaceutical industry demands more than periodic validation and retrospective review.
Why choose AmpleLogic AI APQR Software?
AmpleLogic’s AI-powered APQR solution is purpose-built for pharmaceutical companies, API manufacturers, CDMOs, Biotech, Biosciences, and CROs. This advanced SaaS-based platform is designed in alignment with US FDA and GMP requirements and complies with ICH Q7 (Section 2.5), which mandates periodic quality reviews of APIs, referred to as Product Quality Reviews (PQRs) under EU GMP guidelines.
Intelligent PQR Automation
Streamlines PQR generation using advanced analytics, structured workflows, and end-to-end process tracking to reduce manual effort and improve accuracy.
Continuous CCP Monitoring
Enables real-time monitoring of Critical Control Points with automated alerts for deviations beyond Proven Acceptable Range and Normal Operating Range.
Advanced Sigma Reporting
Generates detailed three-sigma and six-sigma performance reports with dynamic data visualization for deeper process capability evaluation.
Compliance and Quality Oversight
Supports regulatory compliance, faster decision-making, and sustained product quality through real-time notifications and centralized quality visibility.
Challenges with Traditional APQR
Fragmented Data Across Multiple Systems

Manual Consolidation & Spreadsheet Dependency
Reliance on spreadsheets increases manual effort, inconsistencies, and the risk of data errors.

Inconsistent Formats & Review Practices
Lack of standardization leads to variations in APQR structure across products and sites.

Limited Visibility into Quality Trends

Increased Inspection & Compliance
Incomplete traceability and manual records make it harder to respond confidently to audits and inspections.
APQR Designed for Regulated Manufacturing
AmpleLogic’s APQR solution is purpose-built to support regulated manufacturing environments where product quality, data integrity, and inspection readiness are critical.
Don't see your industry? We probably serve it. Let's talk.
Say Hi!Related Articles
FAQ’s
Get Answers to All Your Queries
AmpleLogic APQR is designed to streamline the Annual Product Quality Review process, ensuring compliance, and providing detailed insights into product quality trends.
The software triggers immediate alerts for deviations, allowing timely corrective actions to maintain product quality.
Yes, AmpleLogic offers SaaS (Software as a Service) APQR software specifically designed for the pharmaceutical industry. Our SaaS solution provides a flexible, subscription-based model with automatic updates, ensuring you have access to the latest features and compliance tools at all times. Accessible from anywhere, our software integrates seamlessly with your existing systems, helping you manage product quality reviews efficiently and reduce manual efforts.
Yes, AmpleLogic APQR seamlessly integrates with various systems, including LMS, QMS, BMS, LIMS, Stability, SAP(ERP), DMS and MES/eBMR for comprehensive data capture.
The software conducts statistical analysis on multiple parameters to generate trend charts, control charts, process capability index reports, and Nelson’s rules reports.
Yes, AmpleLogic APQR complies with major standards including 21 CFR PART 11, MHRA, EU Annex 11, etc ensuring regulatory adherence.
The Annual Product Quality Review (APQR) in pharma is a yearly assessment of a product’s quality. It involves reviewing data from manufacturing, quality control, and any issues that arose during the year. The goal is to ensure the product meets quality standards, identify trends, and make improvements, helping to maintain product integrity and comply with regulations.
Pharmaceutical product quality is evaluated systematically in compliance with Good Manufacturing Practices (GMP) through the Product Quality Review (PQR). It involves checking production and quality control information, like records of how products were made, test results, and any problems or complaints that came up. The PQR is used to make sure that products continuously fulfill quality standards, pinpoint areas that need improvement, and guarantee that regulations are followed. This review aids in preserving the general quality, effectiveness, and safety of the product throughout the manufacturing process.
Yes, we offer cloud-based APQR software tailored for the pharmaceutical industry. It enables seamless management of Annual Product Quality Reviews from anywhere, featuring automated report generation, regulatory compliance tools, and secure data storage for enhanced collaboration and accessibility.