Continued Process Verification
Transforming Continued Process Verification into Intelligent, Predictive Quality Assurance
Trusted by 100+ Pharma Companies Globally








Built to Support Modern Regulatory Expectations
Centralized APQR Data Management
Monitor and centralize critical process parameters, quality attributes, statistical trends, and manufacturing performance data within a single CPV platform to ensure ongoing process control and product quality.
Accurate, Compliant, and Audit-Ready Records
Ensure standardized data capture, automated trend analysis, electronic signatures, and comprehensive audit trails to maintain data integrity and comply with global pharmaceutical regulations such as FDA 21 CFR Part 11, GMP, and GxP requirements.
Seamless Cross-Functional Collaboration
Enable quality, manufacturing, validation, and process engineering teams to securely monitor process performance, detect deviations early, and take proactive actions in real time.
Secure, Controlled Audit Access
Provide role-based, read-only access for auditors and inspectors with full traceability, time-stamped verification records, and instant retrieval of CPV data to support smooth regulatory inspections.
Pharmaceutical manufacturing in the modern pharmaceutical industry demands more than periodic validation and retrospective review. As regulatory expectations evolve across the product lifecycle, manufacturers must continuously demonstrate that their manufacturing process remains stable, capable, and in a sustained state of control throughout commercial manufacturing.
Global regulations including Food and Drug Administration 2011 Process Validation Guidance, European Medicines Agency Annex 15, and International Council for Harmonisation Q8, Q9, and Q10 explicitly require ongoing assurance of process performance during routine production. Continued Process Verification (CPV) has therefore become a regulatory and operational imperative for compliance readiness, inspection confidence, and sustained market access.

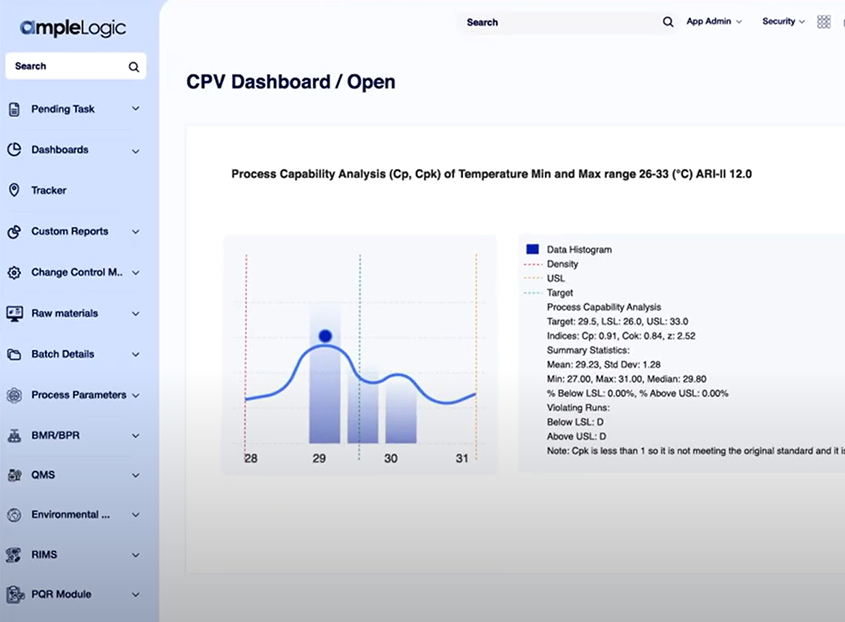
Continued Process Verification enables organizations to continuously monitor critical process parameters (CPPs) and critical quality attributes (CQAs), identify emerging trends within process data, and maintain scientific confidence in production process performance. Effective CPV programs ensure lifecycle process qualification, reduce Out-of-Specification (OOS) events, minimize batch rejections, and support faster, more confident product release.
AmpleLogic CPV is designed as an AI-powered Continued Process Verification and Ongoing Process Verification platform that delivers continuous statistical assurance without dependence on deviation or CAPA execution systems. By combining advanced data analytics, automated statistical tools, robust data connectivity, and explainable AI, it transforms manufacturing data into real-time quality intelligence while preserving full human oversight, regulatory defensibility, and inspection readiness.
Commitment to AI-Powered Excellence
AI-Driven Early Risk Detection
Machine learning detects early process instability using historical and real-time data.

Predictive Process Behaviour Modelling
AI models predict process drift and deviations for proactive control.

Intelligent Signal Prioritization
CPV signals prioritized by risk, severity, and regulatory impact.
Explainable & Audit-Ready Intelligence
AI insights remain transparent, traceable, and support human quality decisions.

Challenges in Manual CPV Processes

Fragmented and delayed data collection
Manual CPV relies on disconnected data from MES, LIMS, historians, sensors, PLCs, and spreadsheets. Lack of integrated data infrastructure delays trend detection and weakens proactive process control.
Limited statistical depth and accuracy
Manual approaches use basic SPC without verifying data distribution. Non-normal datasets analyzed with parametric models weaken validation evidence and regulatory defensibility.

Reactive quality management
Without continuous analytics, instability is identified only after Out Of Specification results, deviations, or batch rejections, increasing investigation effort and compliance risk.

Inconsistent interpretation across sites
Manual CPV depends on individual statistical expertise, leading to inconsistent escalation decisions, governance gaps, and site-to-site variability.

High Resource dependency
Significant QA and manufacturing effort is spent on manual charting and reporting instead of focused scientific interpretation and continuous, data-driven process improvement efforts.

Audit vulnerability
Retrospective evidence compilation limits traceability, delays inspection response times, reduces data transparency, and increases overall regulatory exposure risk during audits.
Challenges with Traditional CPV
Fragmented and delayed data collection

Reactive quality management

High Resource dependency
Key Features of AmpleLogic CPV

Continuous CPP and CQA monitoring
Real-time and historical monitoring across batches, products, and campaigns to demonstrate continuous process control as required by FDA and EMA guidance.

Advanced SPC for normal and non-normal data
Automated distribution identification with correct application of parametric or non-parametric models. Built-in normality tests include Anderson-Darling and Ryan-Joiner.

In-built statistical analysis engine
Validated statistical tools delivering automated univariate and multivariate analysis with full transparency and audit readiness.

Univariate SPC
I-MR, Xbar-R, Xbar-S, and CUSUM charts with configurable Nelson Rules, supported by time-series plots, histograms, probability plots, and capability analysis.

Multivariate SPC
Hotelling T²-based monitoring to detect correlated parameter shifts impacting product quality.

Comprehensive statistical outputs
Mean, median, standard deviation, UCL, LCL, RSD, CV, standard error, range, min/max, Cp, Cpk, Pp, and Ppk with goodness-of-fit evaluation.

Early trend and drift detection
Identification of mean shifts and variability changes before specification limits are exceeded.

AI-assisted predictive insights
Forecasting of drift, capability degradation, and deviation risk to support preventive control.

Batch, campaign, and product-level analytics
Flexible aggregation and drill-down for operational review and enterprise governance.

Audit-ready traceability
Clear linkage between statistical signals, impacted batches, parameters, and timeframes.
Key Enablement Capabilities
Alignment with enterprise Quality Management Systems
Integrated ingestion of manufacturing and laboratory data
Real-time data harmonization and contextualization
Scientifically interpretable statistical outputs
AI-assisted insights with full human oversight
Enterprise dashboards for cross-site visibility
Role-based access for Quality and Manufacturing
Inspection-ready traceability to quality actions
Scalable, multi-site deployment architecture
Why AmpleLogic CPV?
AmpleLogic CPV functions as the analytical nucleus of lifecycle process assurance, delivering continuous and predictive statistical verification while preserving ownership of quality decisions. It strengthens process understanding across process design, process qualification, and commercial manufacturing, ensuring that accountability remains with Quality and Manufacturing teams.
By enabling proactive monitoring, predictive intelligence, and correct statistical interpretation, AmpleLogic CPV shifts organizations from retrospective review to continuous assurance. Early detection of instability reduces OOS events, minimizes batch rejections, improves release timelines, and ensures sustained alignment with FDA Process Validation guidance, EMA Annex 15, and ICH Q8, Q9, and Q10 expectations for ongoing process control.
Industries We Serve
Industry-Specific Software for Enhanced Quality and Compliance
Don't see your industry? We probably serve it. Let's talk.
Say Hi!









